The danger table for hazard Evaluation is crammed out by addressing perfectly-identified threat distinct thoughts for example, “What could go wrong?” Precisely what is the likelihood (probability) that a thing could go Completely wrong?
Regardless that this examine correctly applied QRM, it had some constraints. The primary would be the intensive workload necessary to assess possibility at each and every step of the method. The second limitation is the procedure proprietor’s deficiency of practical experience, which can have an effect on chance assessment.
This agility in launching merchandise don't just provides them a competitive edge but additionally fosters much better relationships with prospects, in the long run leading to greater amounts of satisfaction.
General, aseptic processing transpires in 3 actions that make sure the sterility of items plus the natural environment, as viewed down below.
Every tubing dimension features a tube holder dedicated to exactly just one combination of ID to OD. Shade codes assure exact operation.
These days, a growing range of pharmaceutical brands are working with Sophisticated aseptic processing systems to attenuate operator intervention and contamination possibility from the filling and packaging of liquid parenteral medication.
Terminal sterilization is the whole process of sterilizing solution in its Key packaging utilizing heat or radiation. First, the API and excipients of a formulation are compounded and crammed into vials (or other primary packaging container) and stoppered. The assembled product is then subjected to superior heat or radiation for a click here length of time right up until the product or service is sterilized.
Furthermore, liquid bottle filling devices lead for the mitigation of contamination pitfalls. As a result of their airtight sealing abilities, they protect pharmaceutical items from external contaminants all over their shelf lifestyle.
Validation of component processing should really increase a course of action stimulation examination making use of nutrient medium identified as media fill. A media fill is a single Element of the validation of the aseptic manufacturing process. The aim of the media fill is to check if the aseptic method is satisfactory to prevent microbiological contamination for the duration of the particular approach.
The help provided by all method proprietors and supervisors is considerably appreciated for the valuable feedback and brainstorming classes furnished by in defining hazards inside the cleanroom entry and exit procedures, glass bottle washing equipment operation, and glass filling approach.
Any parenteral solutions plus some implantable equipment read more are the most typical candidates for aseptic processing.
Parenteral merchandise are medications implanted or injected straight with the pores and skin to empower direct administration into tissue organs, blood vessels, or lesions.
If FFS devices are used for the manufacture of non-sterile merchandise, FDAs present Very good Manufacturing Practices (cGMP) necessities must be adopted. When accustomed to manufacture merchandise intended for subsequent sterilization, these devices could be mounted within an setting that will Typically be regarded appropriate for the manufacture and filling of terminally sterilized goods.
This automated procedure allows for the productive and aseptic creation of these containers, decreasing the chance of contamination and making sure the safety and excellent of the ultimate item.
 Tia Carrere Then & Now!
Tia Carrere Then & Now!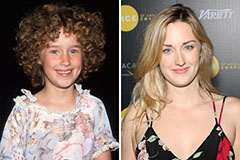 Ashley Johnson Then & Now!
Ashley Johnson Then & Now! Hailie Jade Scott Mathers Then & Now!
Hailie Jade Scott Mathers Then & Now!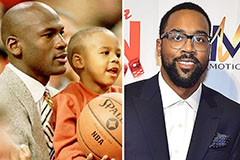 Marcus Jordan Then & Now!
Marcus Jordan Then & Now! Tonya Harding Then & Now!
Tonya Harding Then & Now!